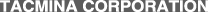Topics|Other What is Gas Lock?
Have you encountered difficulties when injecting sodium hypochlorite for sterilization?
Commonly used as a disinfectant for tap water, swimming pools, public baths, food processors, and a variety of other applications, sodium hypochlorite readily decomposes into a gas. In a diaphragm pump, this can result in a phenomenon known as “gas lock,” which impedes injection of the chemical. TACMINA’s sodium hypochlorite injection pumps feature an innovation that overcomes this issue.
The following describes the basic points for sodium hypochlorite and how gas lock occurs.
What is Sodium Hypochlorite?
Sodium hypochlorite is the chemical compound that results from a reaction between chlorine and sodium hydroxide. It is commonly used as a disinfectant or bleaching agent. Chlorine sterilization with sodium hypochlorite is currently the most common sterilization method in Japan, and it is specified in the Water Works Law. This method provides a more powerful, longer lasting, and lower-cost sterilization effect than is available with other methods.
Because of its strong oxidation properties, sodium hypochlorite has a variety of applications, including deodorization, decolorization, oxidation treatment, deferrization, and demanganization, in addition to sterilization of tap water, swimming pools, and public baths.
Name
- Chemical name
- Sodium hypochlorite
- Chemical formula
- NaClO
- Note : The specific gravity of a 12% solution is 1.2.
Production Method
Sodium hypochlorite is normally produced through a reaction between chlorine and sodium hydroxide.
2NaOH + Cl2 → NaCl + NaClO
(Sodium hydroxide) (Chlorine) (Salt) (Sodium hypochlorite)
- Note : Sodium hypochlorite is typically available on the market in the form of an aqueous solution in the range of 4% to 12% effective chlorine.
Properties
- Sterilization property
- The effective chlorine in a sodium hypochlorite aqueous solution sterilizes bacteria and microorganisms by inhibiting respiratory enzymes.
- Oxidation property
- Generates nascent oxygen, which oxidizes inorganic substances and decomposes organic substances into water and carbon dioxide.
- Bleaching property
- Decomposes colorants through oxidation.
Advantages of Sodium Hypochlorite
Sodium hypochlorite is most commonly used for sterilization of the water supply, swimming pools, public baths, and so on because it is safe and cost-effective and requires no complex facilities.
Benefits
- Highly effective at sterilizing various types of bacteria.
- Safe and easy to handle
- Long-lasting sterilization effect
- The amount of residual chlorine is easily measured.
- Easy to transport and store in liquid form and allows for easy adjustment of the injection volume, resulting in accurate automated control.
- Storage facilities (chemical solution tanks) are easy to install.
Decomposition of Sodium Hypochlorite
Sodium hypochlorite decomposes into salt and oxygen by the action of sunlight (ultraviolet radiation) and high temperatures.
- Photochemical decomposition
- Sunlight, especially ultraviolet radiation, accelerates decomposition. After about 20 hours of exposure to sunlight, 90% of the effective chlorine decomposes into salt and oxygen.
- Heat decomposition/natural decomposition
- Decomposition can be accelerated by increase in temperature.
(Sodium hypochlorite) (Salt) (Oxygen)
The Gas Lock Phenomenon in Diaphragm Pumps
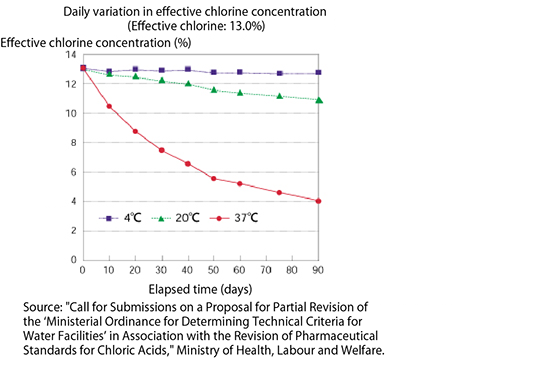
Diaphragm pumps are generally used for the injection of sodium hypochlorite because they maintain accurate injection volumes and are reliable for injection and easy to maintain. A diaphragm pump feeds liquids through the reciprocating action of the diaphragm, (a rubber or Teflon membrane), which varies the inner volume of the pump head.
- Note : For information on the operating principle of a positive displacement pump, see “Basic Course 1-1. What is a positive displacement pump?”
Because sodium hypochlorite readily forms a gas, using a diaphragm pump to inject sodium hypochlorite can result in the following problems.
- (1) The gas enters.
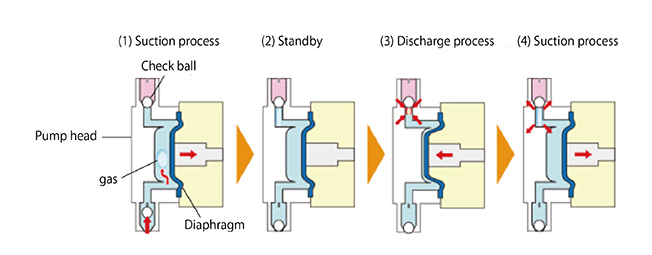
- (2) The gas accumulates in the upper interior of the pump head.
- (3) The gas is compressed in the pump head, but no liquid is discharged.
- (4) The compressed gas returns to its original volume, but no liquid is drawn in.
If gas enters the pump head, the check ball on the discharge side acts as a lid. Only the gas expands and compresses, but no liquid is discharged at all, even with a reciprocating diaphragm.
This describes the "gas lock" phenomenon.
How to Recover a Diaphragm Pump From Gas Lock
Any of three methods can be used to discharge the gas and overcome gas lock.
- 1.Discharge the gas manually.
- Open the air vent valve on the pump head to discharge the gas. Although manual gas discharge is easy, it must be repeated every time gas lock occurs.
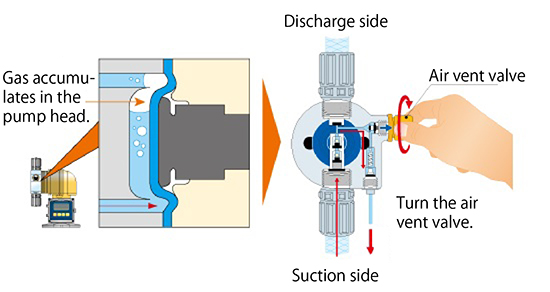
- 2.Discharge the gas by increasing the compressibility of the air.
- Increase the compressibility by increasing the volume discharged in a single evacuation, thus easily discharging the gas in the pump head. However, this method is ineffective when a large amount of gas is present.
- 3.Install an automatic air vent valve to discharge the gas.
- Branch the pipe on the discharge side of the pump into a chemical feed pipe and an air vent pipe. Gradually discharge the gas while feeding the chemical solution. With this method, however, gas as well as a large amount of chemical solution flow into the air vent pipe when the pressure rises in the chemical feed pipe, resulting in unstable injection.
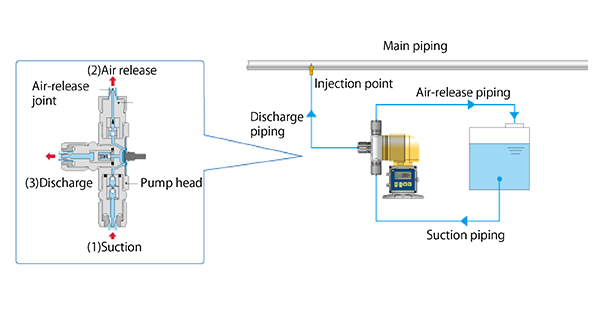
- (1) Suction
- Generated gas is drawn into the pump head.
- (2) Discharge of gas
- Only the drawn-in gas returns through the air vent valve to the chemical solution tank.
- (3) Discharge
- After the gas is discharged from the pump head, sodium hypochlorite is fed to the discharge pipe through the backpressure valve and joint on the discharge side.
TACMINA Anti-Gas Lock Sodium Hypochlorite Injection Pumps
TACMINA offers the DCLPW and CLPW Series of pumps that are specially designed to inject sodium hypochlorite.

Video of CLPW Automatic Gas Discharge Mechanism
The following video demonstrates the operation of the mechanism.